Future Horizons:
10-yearhorizon
Rejuvenation becomes safe
Livers, hearts and kidneys are transplanted after perfusion with epigenome-modifying agents that rejuvenate the organs. In vivo reprogramming of specific pathologies is tested on debilitating single-tissue pathologies for which risk is tolerable, for example COPD, chronic kidney disease and rheumatoid arthritis.
25-yearhorizon
In vivo rejuvenation becomes widespread
In vivo reprogramming targets normal human ageing. Research identifies a master cell type that systemically rejuvenates tissues, enabling new forms of intervention.
Epigenetic reprogramming to turn the body’s somatic cells into pluripotent cells has been investigated for decades. This is mediated by proteins called transcription factors and has been shown to have rejuvenating effects in preclinical trials.6 However, the mechanism (which uses four transcription factors — known collectively as OSKM — that are delivered by viral payload) carries an inherent risk of cancer.7 Safety will be key to human translation, meaning that potentially cancer-causing transcription factors will need to be replaced by small molecule drugs or other alternatives. Early work identifying safer transcription factors, including HNF4 alpha, which can rejuvenate the liver, is under way. The epigenome may also be targeted more indirectly, including by the SIRT6 activator fucoidan or forskolin.8
Instead of in vivo modification, ex vivo interventions could be undertaken to reprogram aged stem cells before reintroducing them to the body. This would allow more control of the process. This approach is a focus of current research for reprogramming transplant organs, such as kidneys and hearts, which are epigenetically rejuvenated before being introduced into a recipient.9
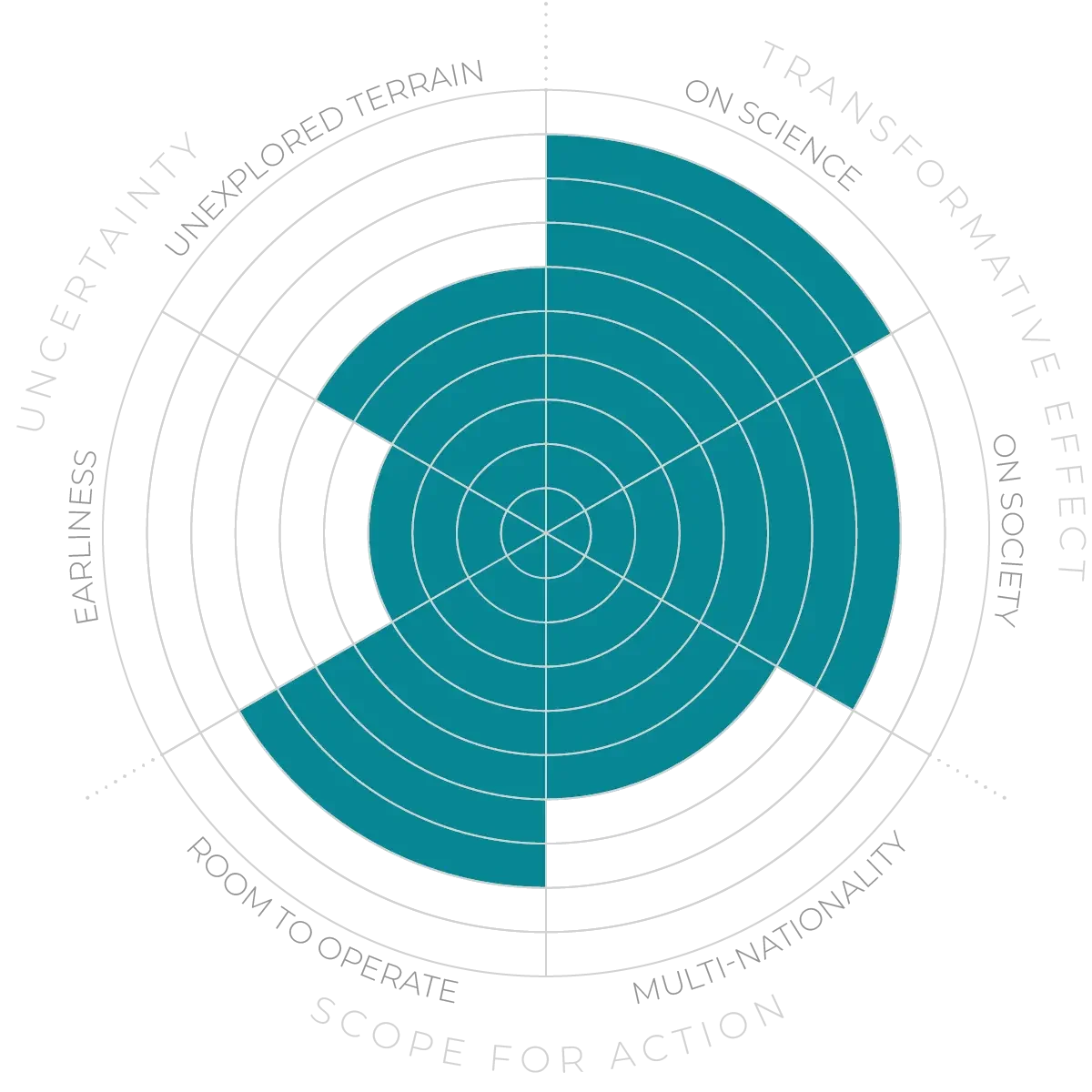
Rejuvenating the epigenome - Anticipation Scores
The Anticipation Potential of a research field is determined by the capacity for impactful action in the present, considering possible future transformative breakthroughs in a field over a 25-year outlook. A field with a high Anticipation Potential, therefore, combines the potential range of future transformative possibilities engendered by a research area with a wide field of opportunities for action in the present. We asked researchers in the field to anticipate:
- The uncertainty related to future science breakthroughs in the field
- The transformative effect anticipated breakthroughs may have on research and society
- The scope for action in the present in relation to anticipated breakthroughs.
This chart represents a summary of their responses to each of these elements, which when combined, provide the Anticipation Potential for the topic. See methodology for more information.